
Introduction
The successful development of biotherapeutics requires an in-depth understanding of the impacts associated with mechanical and physical stress. Instability and subsequent aggregation can render them biologically inactive or even induce an immunological response in patients. Stirring stress is one of the most notable factors which disrupts native biopolymer structure and promotes aggregation. However, contact stirring represents only one type of stress that can occur during the purification, manufacturing and packaging of therapeutic proteins. Overhead stirring, capillary shear stress, filtration, and peristaltic recirculation can all impact protein stability and induce aggregation. Since many or all of the listed mechanical and physical stressors can be present during therapeutic protein production, it is critically important to quantitatively assess the impact of each stressor.
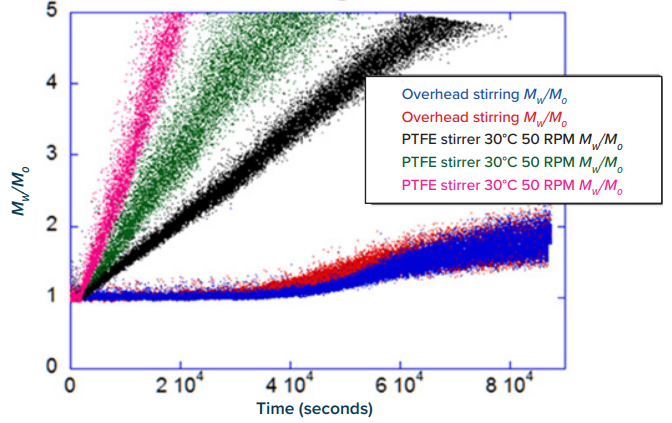
ARGEN™: Smart & Rapid Therapeutic Biopolymer Development ARGEN™ is a high throughput tool for the rapid assessment of the stability and viability of therapeutic proteins, peptides, and biopolymers. The instrument uses a multi-stressor testing platform powered by static light scattering detection and intuitive data processing. These features enable teams to develop biologic formulations up to 16-fold faster. How ARGEN™ Works ARGEN™ utilizes fixed angle (90°), simultaneous multiple sample light scattering (SMSLS) technology which provides rapid, real-time, continuous data collection for characterizing qualitative and quantitative properties of target molecules. The device is equipped with 16 independently controlled sample cells, permitting the user to establish thermal, chemical, and mechanical (stirring) stress parameters on each sample concurrently. This allows for a highly flexible approach to experimental design. ARGEN™ Intuitive Control Software The ARGEN™ control software features an intuitive interface for all aspects of experimental design and independent control of each cell for parallel parameter adjustment and real-time data processing. Understanding the impacts of different modes of stirring stress provides insight into the stability of therapeutic biopolymers and their ability to remain stable during purification and manufacturing processes. Each cell of ARGEN™ is equipped with a stepper motor attached to a rotating magnet which couples to a micro stir bar either at the bottom of the cuvette (contact stirring) or overhead (non-contact stirring) to simulate perturbations during bioprocessing. Stirring/agitation rate (0-2000 RPM) is manually controlled for precision via the ARGEN™ intuitive control software. Overhead stirring is performed with a custom engineered cuvette. Simply, the cap of the cuvette is fitted with a spindle which suspends a micro stir bar. Sealed bearings housed in the cap allow the spindle and stir bar assembly to freely rotate in sync with the magnetic stepper motor. Figure 1 demonstrates the difference in aggregation rates (slope of Mw/Mo) between contact stirring and non-contact stirring. Changes in Mw/Mo and aggregation were observed at the outset of all (triplicate) contact stirring experiments. Whereas the protein remained stable for approximately 10 hours when subjected to non-contact stirring, this data clearly provides evidence that changing the mode of stirring stress applied to the monoclonal antibody results in very disparate aggregation rates and profiles which can be vital during the vetting process and identifying the most viable construct. Understanding the impacts of stirring and shear stress on biopolymers during processing and manufacturing are key to expediting development. The ability of ARGEN™ to simulate stirring stress while simultaneously monitoring oligomerization or degradation states is unique and provides the final link in getting a treatment to market. This case study provides evidence of the effects of different modes of stirring stress experienced during bioprocessing on a monoclonal antibody. Monitoring changes in normalized molecular weight in real time demonstrated that contact stirring would not be a preferred method as the protein aggregated immediately. This data is critical in vetting constructs to discover the optimal candidate as well designing processing equipment to minimize the impacts and perturbations experienced during bioprocessing.
Characterizing the Impacts of Different Modes of Stirring Stress
Conclusion


